Surveying Wood Hulls
Part 1: Materials and Causes of problems
by David Pascoe, Marine Surveyor
Introduction - Part I - Part II - Part III
Materials
Before getting into the heart of the matter, lets first consider the nature of the materials that we are dealing with, for this will give us a far better understanding of why severe problems frequently go undetected.
Contents
Materials:- Wood
- Metals
- Electrolysis
- Galvanism
- Stray Current
- Oxygen Starvation
- Climate
- Hull Stress
- Wood and Water
Corrosion:
Other Factors:
Wood is a natural, organic material that has no
consistency from one species to another, or within a species. Each tree
grows differently and yields different qualities of wood. Consistency may
or may not exist. There are literally hundreds of varieties of species
wood used in construction, and these species also have an equally wide
range of strength and other characteristics. Of all these species, only a
very few have the ideal characteristics of good strength and resistance to
deterioration. (here deterioration means fungicidal micro-organisms and
electro-chemical degradation). Woods like teak, cypress, fir, long leaf
yellow pine, mahogany and only two of hundreds of varieties of oak fit the
bill. But over several thousand years of boat building, these most
desirable species have been consumed and all but disappeared, causing
builders to have to utilize ever lower grades of material.
"Wind shakes" are a phenomenon caused when a tree is hit by high winds,
causing the whole trunk to twist. This can damage the wood in ways that
aren't easily detected. It causes minute splits in the fibers which will
make the wood more porous and cause a plank cut from that tree to rot much
quicker. This is why we often find just one plank on the whole vessel that
rots badly with no apparent explanation for why.
Teak and cypress, among others, are highly valued for their fungi
resistance. That resistance comes from certain chemicals in the tree that
are toxic to fungi (and man as well). These trees can also grow in soil
where those chemicals are absent, meaning that they will not be present in
the wood and therefore not be rot resistant. This also explains why rotten
planks and frames will appear in places where it is not expected.
Strength of Wood is degraded by a variety of factors: soaking in
water for 10, 20, 30 or more years, micro-organisms, shipworms, ants,
termites, the normal stresses imposed on the hull, sunlight, constant
wetting and drying and chemicals introduced into the interior of the hull
such as spilled battery acid, petrochemicals, detergents and chlorinated
cleansers, etc.; all work to take their toll on wood. In other words,
unlike materials such as fiberglass and aluminum, wood is degraded by a
large variety factors.
Metals Wood construction, of course, must
utilize some sort of metal fasteners to hold it together. As with the
quality of the wood, the quality of the fasteners used in the trade can
run from very poor (steel) to fairly good (silicone bronze) to excellent (monel).
As with everything else in this world, the higher the quality, the more it
costs. Thus, the ever-present tendency to reduce quality to reduce the
cost of the product. This is one thing the surveyor should never loose
sight of, for it can also come back to bite him if he does.
Metals , of course, are all subject to corrosion, a phenomenon
which is subject to much myth and little understanding. In industry,
corrosion is still one of the most highly studied areas of metallurgy.
Even after billions of dollars of research, all of its causes and effects
are still not fully understood. So when we think that we have some
understanding about the nature of corrosion, we should think twice.
The final consideration of materials leads us to consider how they all
work together, and it is here where the general understanding of wooden
vessels usually falls short.
Corrosion
Corrosion is a natural process that works to degrade all materials, whether its gold, rock, plastic or steel. Corrosion occurs in many forms, most of which are not common knowledge, even in the marine industry. The ones we experience most often are oxidation reduction, galvanism and electrolysis. I will start with the last first because it is so misunderstood.
Electrolysis is the same identical process used in electroplating. It occurs only with DC current and essentially moves material from an anode to a cathode. Theoretically, electrolysis cannot occur from AC current, although in reality stray AC current can be converted to DC current through naturally occurring diodes such as salt crystals. That's why electroplating can only be done with the very expensive process of using DC current. Otherwise, plating would be cheap and not as expensive as it is.
Electrolysis only occurs on boats when there is a ground fault and it is leaking current, usually from bad wiring. Small current leaks are commonplace and are rapidly dissipated in surrounding metals without doing damage. Larger current leaks, as when a significant part of a large conductor has a ground fault, can produce very damaging corrosion. People often see heavy pitting on propeller blades and rudders and attribute this to electrolysis when, in fact, it is usually the result of yet another form of corrosion, erosion corrosion caused by fast streams of water laced with air bubbles. Erosion corrosion is strictly a mechanical form of corrosion.
Galvanism This is a from of corrosion caused by a current flow generated between two materials with greatly differing electrical potentials. Contrary to popular belief, galvanism does not occur just between two different metals; it can occur between other materials, such as carbon rubber and a metal. Carbon rubber mated against aluminum, for example, is a disastrous combination that will destroy aluminum very quickly. This is often found in carbon rubber hose joined to aluminum or copper pipes and is often referred to as electro-chemical degradation (ECD).
On wooden boats, galvanism is rarely a factor except in small isolated areas. When two dissimilar metals are mated electrically, they generate a very small amount of current that has little power. That means that the electricity generated has little capacity to flow very far before being dissipated. Wood is an extremely poor conductor, and sea water wet wood is not much better, although it will conduct current for short spans before being generally dissipated. For this reason, stray current in a wooden hull does not have the capability to generally damage hull fasteners, even when underwater metals are bonded. When zincs are attached to the system, all potential for galvanism is eliminated so long as the zincs remain effective.
Bottom fasteners, of course, are not normally exposed to water, and thus their chance of being affected by stray current or galvanism is even further reduced. Thus, galvanism rarely, if ever, is a factor in hull factor corrosion.
Stray Current is the cause of true electrolysis and can cause damage to fasteners if the leaking voltage is high enough, has sufficient power flow (amperage) and continues long enough under ideal conditions. However, I have never found a case where stray current damage to fasteners was not also accompanied by severe corrosion damage to exposed, submerged metals such as propellers, shafts, sea cocks and the like. Absent such damage to exposed metals, there is no reason to suspect stray current damage to fasteners as fasteners are the last to become damaged in the chain of events.
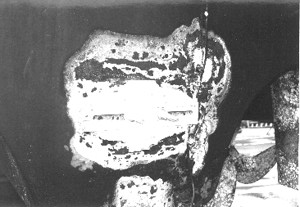
The effects of stray current are usually unmistakable. This is the remnant of a zinc anode attached to a bonding system that was subjected to a full twelve volts, leaving an obvious burn pattern. The zinc did its job and the problem was caught in time before serious damage occurred.
Electrolysis, or stray current corrosion is usually notable by the very
bright appearance of the corroded metal. When the pitted part of the metal
is bright, that means that the corrosion is so rapid that oxides have no
chance to form. That's particularly true with all copper-based alloys like
brass and bronze. When the surface is dull, but still brightly colored
such as pink or orange, this tends to indicate a less rapid rate of
corrosion, but stray current nevertheless.
Oxygen Starvation This is the primary
cause of corrosion to hull fasteners, also known as crevide corrosion.
Oxygen starvation is not the simple form of chemical reaction with water
that causes rust or other surface oxides, but the result of water being
trapped in a small, confined space where the oxygen supply is cut off.
Surveyors are familiar with this phenomenon on aluminum fuel tank bottoms
mounted on a wet plywood deck that results in rapid and severe corrosion
pitting. What most surveyors have never understood is that this same
phenomenon occurs with metal fasteners joining two pieces of wood
together, or any other material for that matter.
By the time a surveyor has pulled a thousand bronze screws out of hull
bottoms, it becomes readily apparent that the wastage of the screws, or
necking as its sometimes called, always starts at the center of the screw.
This coincides with the interface between the plank and frame. The reason
it does is because the seam between plank and frame is subject to the
capillary effect which, as we know, is the tendency of water to follow
along cracks, seams or fissures. Therefore, anytime the plank/frame
interface is wetted, the water is going to head straight for the screw.
Now, the screw, being spiral threaded, creates the tendency for that water
to follow the treads right up and down the shank of the screw. Occasional
wetting of the fastener is not, of course, a problem in and of itself. But
once the fastener is wetted a few times, it acquires a slight layer of
soft and very porous oxides. Normally, these oxides would serve to protect
the screw from further corrosion, but something else interferes. The
oxides on the screw surface further enhance the capillary effect the same
way that beach sand absorbs water. Thus, once the corrosion process
starts, it can only accelerate.

Corrosion of stainless bolts by crevice corrosion or acid attack. The prominent feature is that the active corrosion areas are bright metal.<
Unfortunately, yet another factor gangs up on our poor fasteners: crevice corrosion. The water entrapped within the screw cavity or interface between the planks does not have a good oxygen source. i.e., air flow. The chemical reaction of oxidation of the metal robs the water of oxygen and turns the water to an acid. So what we now have is first, oxidation followed by acid attack which really does the most damage. These processes are endlessly repeated over the years, at an ever accelerating rate until the fastener is finally destroyed.

This is erosion corrosion on a rudder blade caused by propeller wash. It is often mistaken for electrolysis.
The feature that often leads surveyors and investigators to the conclusion of galvanism stems from the fact that oxidation reduction or acid corrosion usually leaves the eroded metal bright-looking and without the usual covering of granular oxides. Based on what we usually see of galvanic or stray current corrosion, this seems like a reasonable conclusion but its not. Acid corrosion is usually recognizable by pink and black residues on a bronze fasteners. On a steel fastener it tends to be very black. If there is no black but just pink residue on the bronze fastener, then water is getting to it on a continuous basis.
Other Factors
Climate The effect of climate on wood vessels
simply cannot be understated. In the Baltic sea archaeologists have been
recently pulling up very well preserved parts of wooden vessels that are
600 to 1,000 years old. In the tropical waters of Florida, vessels sunk as
little as 60 years ago have completely disappeared. Of the many Spanish
galleons of the silver fleet sunk on Florida shores 300 years ago, almost
no wooden parts remain. Such is the effect of warm, tropical waters on
wood. Cold water, of course, has a preservative effect on both wood and
metal.
The other great enemy of wood are very damp and rainy climates, especially
moderate and tropical climates. In itself, salt is a preservative because
fungi can't survive in a salt environment. But nothing advances fungicidal
attack on wood like nice, fresh rain water. Of course, one of the greatest
maritime myths of all time is the notion of "dry rot." Wood does not rot
unless it has a source of water. Fungi does not live, and wood does not
rot in the desert. Therefore, there is no such thing as dry rot.
Fresh water often does its worst damage when it leaks into a hull. This
happens in several ways. First, water from the decks runs down the sides
of the hull and gets absorbed into open seams in the planks. Second, deck
leaks run down the inside of the hull where the same thing happens, but
also ends up laying in the bilge where there is often a perfect
environment of nutrients for fungi to blossom.
The problem for surveyors is that boats don't stay put and you don't know
its past history and where it came from. Therefore, one has to expect the
worst.
Hull Stress The deep bilge of course is always
the first place the surveyor looks for bad fasteners, for it is here where
there is a constant source of water. Yet boats are not monolithic
structures and they are subjected to stress and thus work and torque and
wrack and twist in all directions. Often not enough to be visually
detectable, but enough that all the seams and joints are indeed moving.
Projected over thousands of cycles over the years, this creates the
potential source for water entering virtually any seam in the hull bottom,
not once but hundreds of times.
Any surveyor who has spent a significant part of his career surveying wood
boats knows that, other than the garboards, wasted bottom fasteners can
occur anywhere, and just about any point in the life span of the vessel.
He knows its not reasonable to apply any particular time limit, for it is
entirely dependent on whether open seams are allowing water to get at the
fasteners.

Hogging, a condition in which a hull is actually sagging with age, is an accurate indicator that a wood hull has reached the point of severe structural degradation and can no longer be considered safe. This condition is plainly evident on the above yacht
Wood and Water The structure
of wood is made up of very oblong cells of cellulose fiber. Living wood
conducts water soluble nutrients along the grain of the wood, or from
end of cell to end of cell, ultimately to the top of the tree. Wood does
not transmit water across the grain very well at all, either from the
edge or the wide surface. We have only to stand up a plank in a pan of
water to understand how well this works. Very soon, the plank will empty
the pan of water.
Because a plank is no longer a living organism, it has lost much of its
ability to transmit water along its length. Eventually it reaches what is
known as equilibrium level and will absorb no more. Complete saturation of
the open cells will only occur a short distance from the end grain unless
rot sets in and then it will advance further.
When it comes to fasteners, the effect of this point should be rather
obvious: Fasteners near the ends of planks are the ones most
vulnerable. (next)
- Introduction
- PART 1: Materials and Causes of Problems
(with 4 pictures)
Materials: Wood - Metals, Corrosion: Electrolysis - Galvanism - Stray Current - Oxygen Starvation, Other Factors: Climate - Hull Stress - Wood and Water - PART 2:
How to Survey A Wood Hull
Opening Up - Planks & Frames - Weepage - Chine Areas - Forefoot - Transom - Keel, Bolts, Keelson, Clamps and Stringers - Inaccessible Areas - Fuel & Water Tanks - The Exterior - The Bottom Survey - Steel Fasteners - Screw Fastened Vessels - Acceptable Degree of Wastage - Summary of Structural Strength - PART 3: Appendix
[A] Hull Planking Types
[B]Recommendations for Safety Equipment and Systems
Professional Marine Surveys
- Hull Design Defects - Part I
- Hull Design Defects - Part II
- Surveying Boats With Molded Integral Grid Systems
- Surveying Wood Hulls
- Avoiding the Blister Blues
- Sinking of EL TORO II
- Storm Damaged Boats
- Insurance Surveys and Reports
Power Boat Books
 Mid Size Power Boats
Mid Size Power Boats A Guide for Discriminating Buyers
Focuses exclusively cruiser class generally 30-55 feet
With discussions on the pros and cons of each type: Expresses, trawlers, motor yachts, multi purpose types, sportfishermen and sedan cruisers.
Selecting and Evaluating New and Used Boats
Dedicated for offshore outboard boats
A hard and realistic look at the marine market place and delves into issues of boat quality and durability that most other marine writers are unwilling to touch.
2nd Edition
The Art of Pre-Purchase Survey The very first of its kind, this book provides the essentials that every novice needs to know, as well as a wealth of esoteric details.
Pleasure crafts investigations to court testimony The first and only book of its kind on the subject of investigating pleasure craft casualties and other issues.






